
Figure 1 from Screening of Yeasts for Cell-Free Production of (R)- Phenylacetylcarbinol in a Shake Flask Condition | Semantic Scholar
![PDF] The yeast mediated synthesis of the l-ephedrine precursor, l- phenylacetylcarbinol, in an organic solvent | Semantic Scholar PDF] The yeast mediated synthesis of the l-ephedrine precursor, l- phenylacetylcarbinol, in an organic solvent | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/96025f6077bf82ffa0b924f17d008ebacae4a94e/22-Figure1.1-1.png)
PDF] The yeast mediated synthesis of the l-ephedrine precursor, l- phenylacetylcarbinol, in an organic solvent | Semantic Scholar
Selection of Yeasts for the Production of L-phenyl-acetil- carbinol Bybiotransformation in Shake Flasks

Asymmetric synthesis of ( S )-phenylacetylcarbinol – closing a gap in C–C bond formation - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC01803C
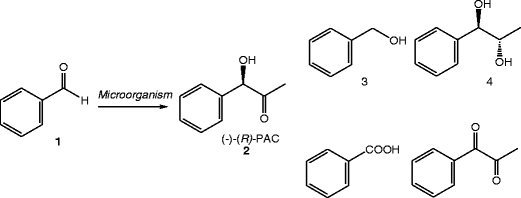
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

Stereoselective synthesis of (1R, 2S)-norephedrine by recombinant whole-cell biocatalysts coupling acetohydroxyacid synthase I and ω-transaminase - ScienceDirect

An N-methyltransferase from Ephedra sinica catalyzing the formation of ephedrine and pseudoephedrine enables microbial phenylalkylamine production - ScienceDirect

Biotransformation of benzaldehyde to L‐phenylacetylcarbinol (L‐PAC) by Torulaspora delbrueckii and conversion to ephedrine by microwave radiation | Semantic Scholar
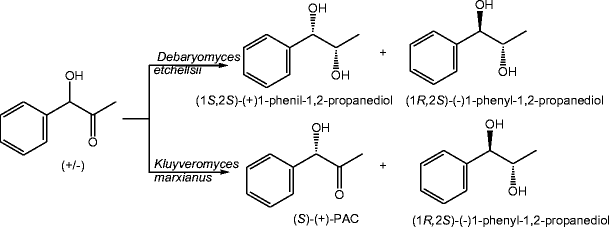
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink








