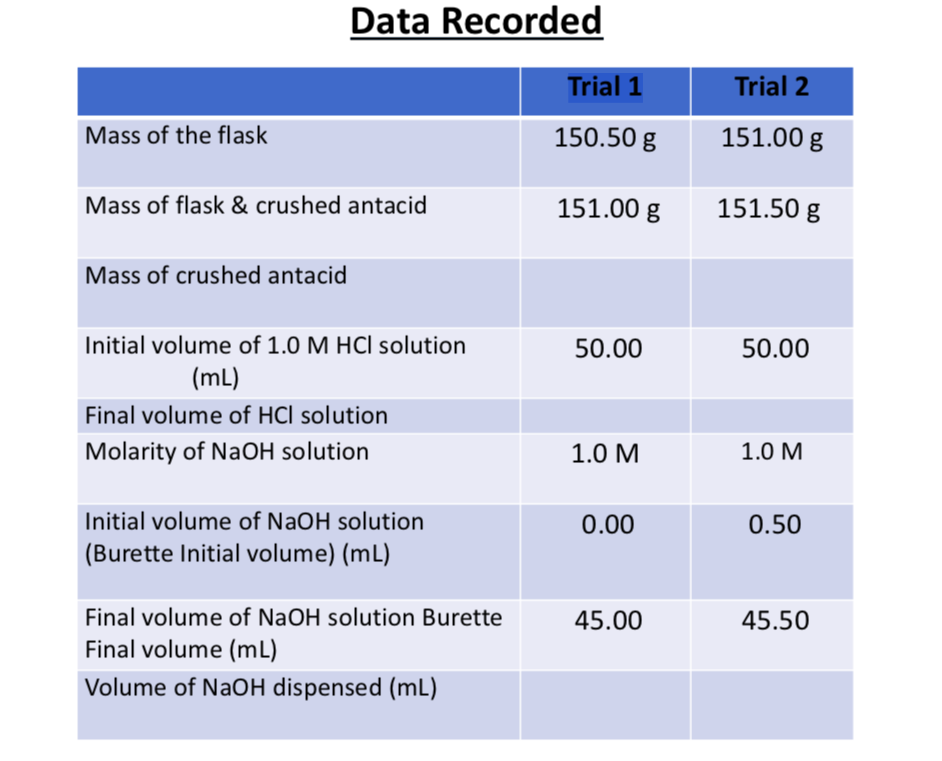
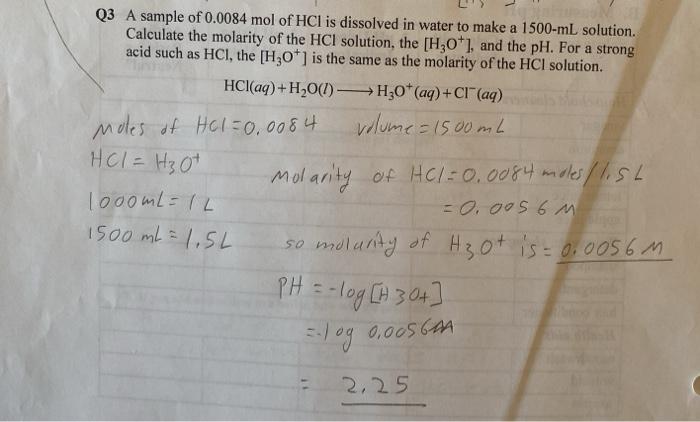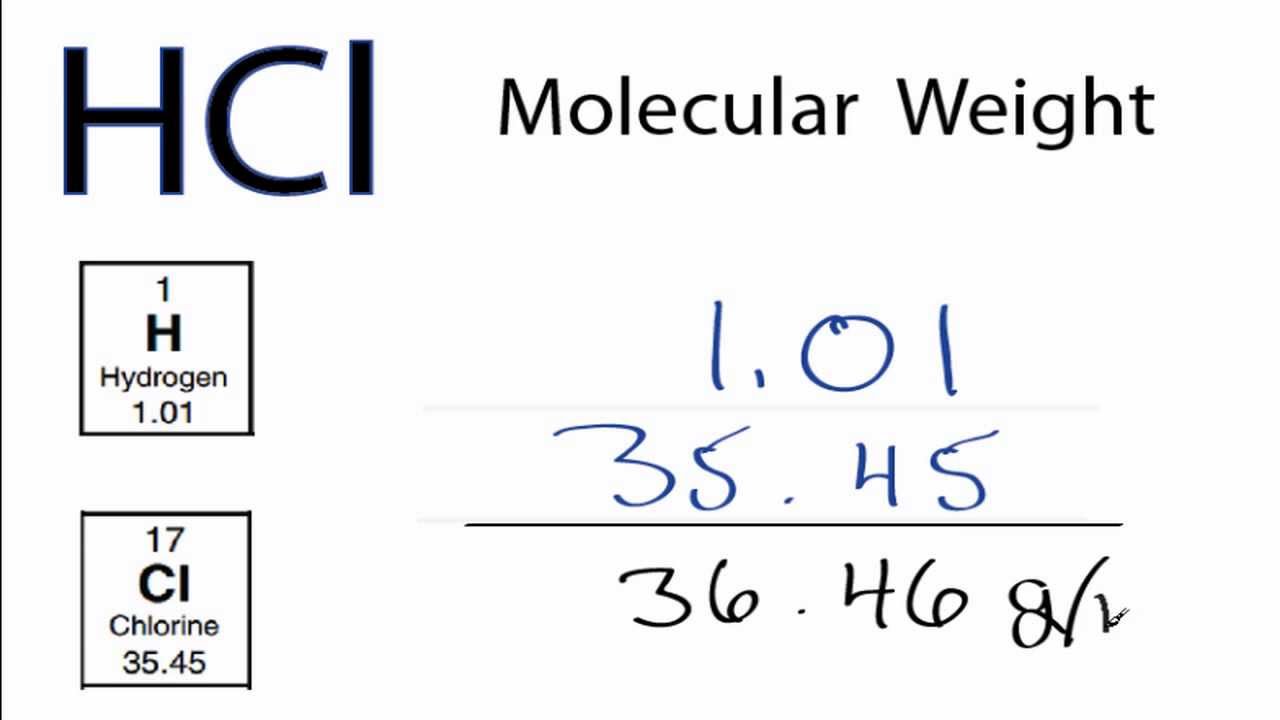One liter of 5.0 M HCl contains how many moles of HCl? M = 5.0 mol= ? L = 1 L x = 5 mol. - ppt download

SOLVED: Q : Hydrochloric acid (FW=36.46 g/mol) is sold as 37% ww hcl with a density of 1.18 g/ml: The value of the solution 0.315mol of hcl is 26.3 ml 20.0 ml 2.0 ( 10.0 ( 2.6 ml ehogkky?

SOLVED: Sample Exercise 13.6 Calculation of Mole Fraction and Molality An aqueous solution of hydrochloric acid contains 36% HCl by mass. (a) Calculate the mole fraction of HCl in the solution. (b)
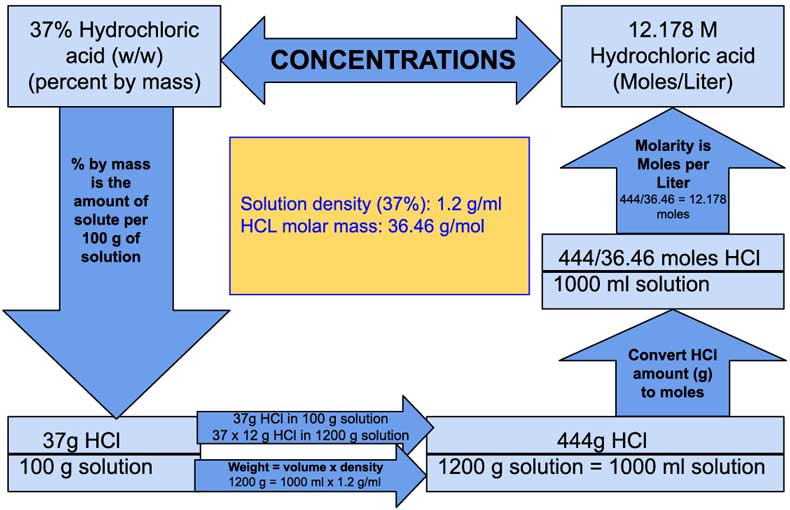
How to Calculate Molarity of 37% (w/w) Hydrochloric acid (HCl), Alternative Method - Laboratory Notes

SOLVED: Sample Exercise 13.6 Calculation of Mole Fraction and Molality An aqueous solution of hydrochloric acid contains 36% HCl by mass. (a) Calculate the mole fraction of HCl in the solution. (b)

Hydrochloric Acid, c(HCl)=0.1 mol/L (0.1N) Titripur , MilliporeSigma, Quantity: 1 L | Fisher Scientific

SOLVED: 1. A solution of hydrochloric acid contains 36% HCI by mass: A Calculate the mole fraction of HClin the solution: B Calculate the molality of HCl in the solution. 2.A solution

Question Video: Calculating the Concentration of a Hydrochloric Acid Solution Using Experimental Data | Nagwa