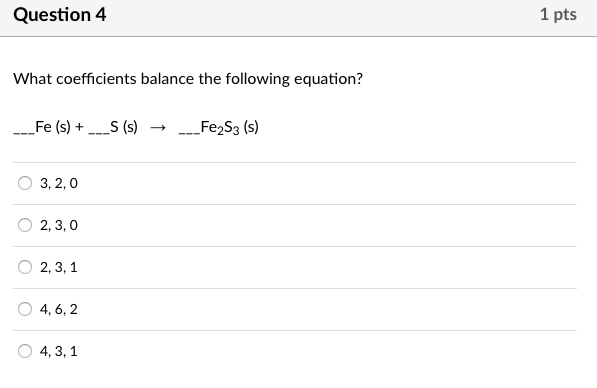
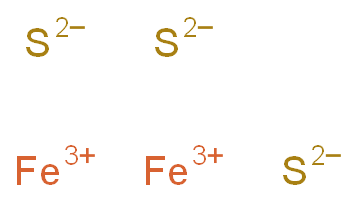SOLVED: Balance the equation Fe2S3(s) → Fe(s) + S(s) + 69kJ 1b) 2A. How many moles of sulfur are in one mole of Fe2S3(s)? 2b. How many moles of Sulfur is in

for the reaction fe2s3+5o2gives raise to 2feso4+so2 the equivalent weight of fe2s3 is - Chemistry - Solutions - 13840395 | Meritnation.com
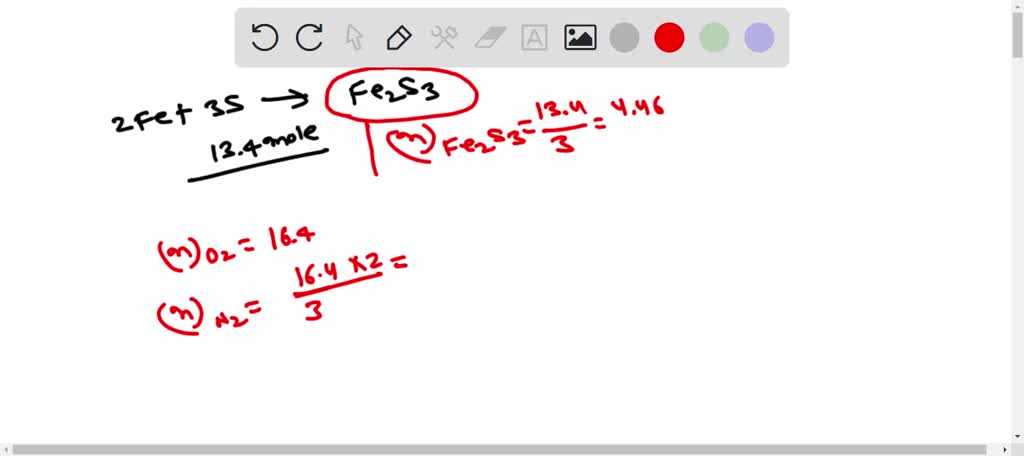
SOLVED: 1. How many moles of Fe2S3 can form from 13.4 moles of S? 2Fe +3S –> FE2S3 A. 40.2 Moles B.4.47moles C.0.224 moles D.13.4 moles 2. How many grams of N2
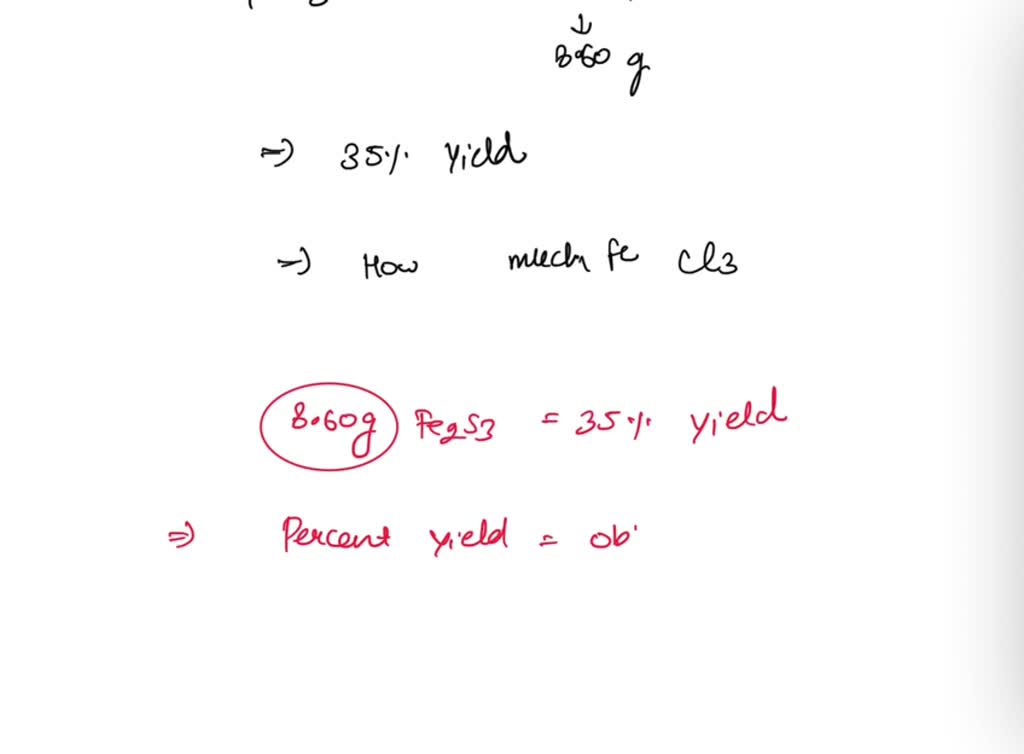
SOLVED: A student decided to prepare Fe2S3 by reacting FeCl3 with H2S. The other product was HCl. The student collected only 6.20 grams of Fe2S3. The notebook indicated that the expected percent