
Calculations in Chemistry To calculate the number of moles in a solid we use the following Mole Triangle g n gfm g = Mass in Grams n= Number of moles. - ppt download
The CV (a) and CA (b) curves for the electrochemical oxidation of 0.5... | Download Scientific Diagram

SOLVED: An ideal gas has a constant molar specific heat cv= 564 J/K/mol. Calculate the change in entropy per mole in going from the state (V1,T1)=(12 m^3,19 C) to the state (V2,T2)=(551

Moles and Solutions g n gfm To calculate the number of moles in a solution we use the following n CV n = number of moles C = concentatration (mol/l) V. - ppt download
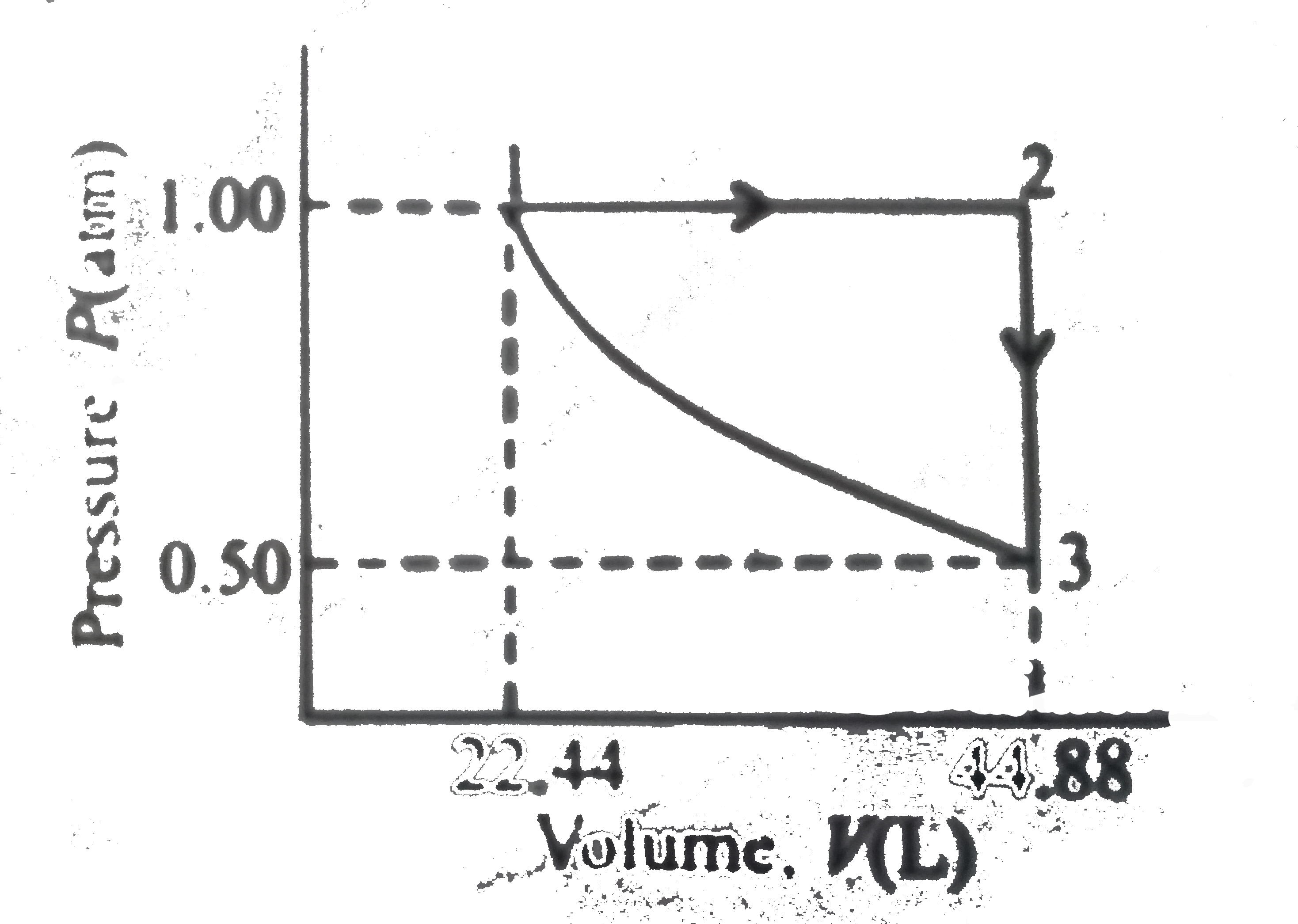
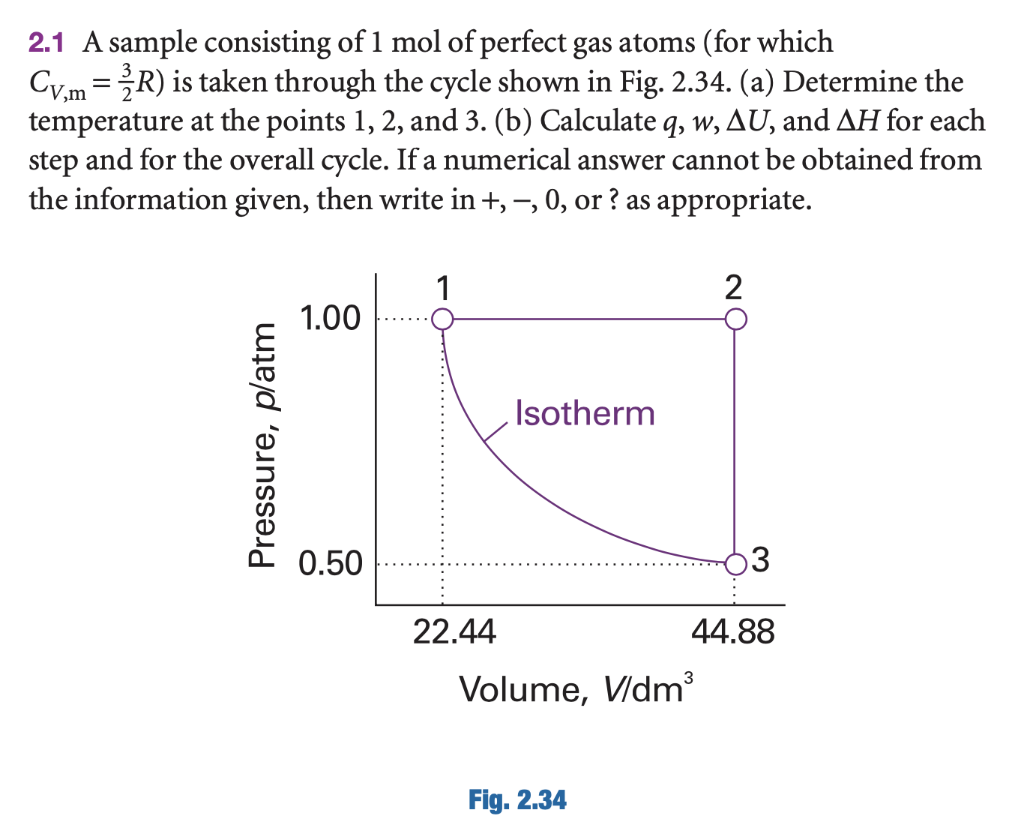
A sample consisting of 1mol of a mono-atomic perfect gas (C(V) = (3)/(2)R) is taken through the cycle as shown. Temperature at points (1),(2) and (3) respectively is

Moles and Solutions g n gfm To calculate the number of moles in a solution we use the following n CV n = number of moles C = concentatration (mol/l) V. - ppt download

A) CV curves of 0.04 mol L À 1 BR buffer (pH 4.0) with 5 % of DMSO (À... | Download Scientific Diagram

Calculate the value of gamma = Cp / Cv for a gaseous mixture consisting of v1 = 2.0 moles of oxygen and v2 = 3.0 moles of carbon dioxide. The gases are assumed to be ideal.
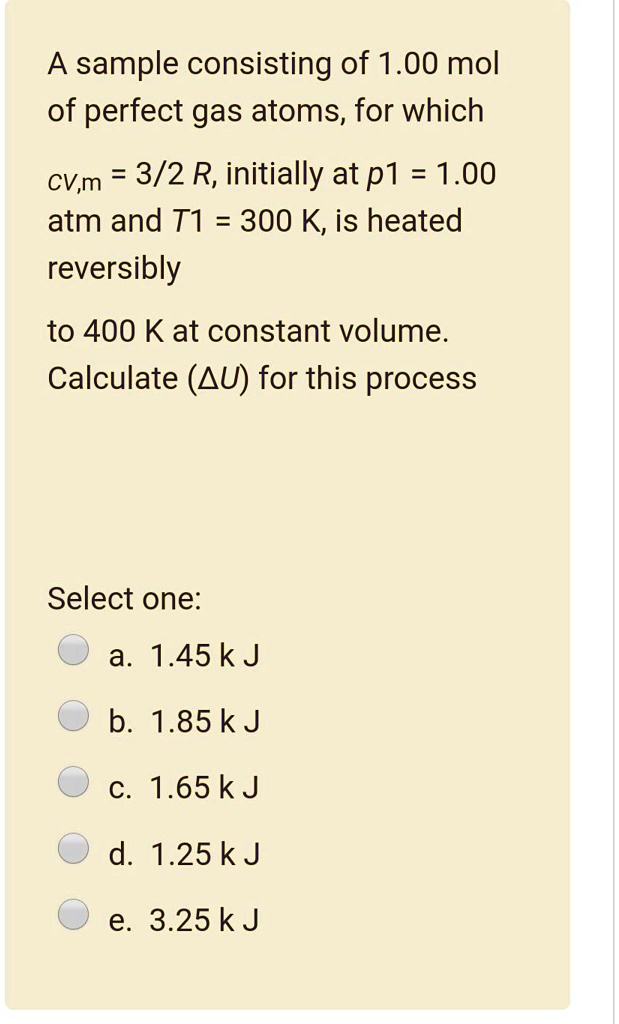
SOLVED: A sample consisting of 1.00 mol of perfect gas atoms, for which CVm = 3/2 R, initially at p1 1.00 atm and T1 = 300 K, is heated reversibly to 400

A reversible cycle executed by 1 mol of an ideal gas for which Cp = (7/2) R and Cv = (5/2) R consists of the following: a. Starting at T1 = 600


![N=CV, CONCENTRATION, VOLUME, NUMBER OF MOLES [Last minute revision] | Chemistry at glance - YouTube N=CV, CONCENTRATION, VOLUME, NUMBER OF MOLES [Last minute revision] | Chemistry at glance - YouTube](https://i.ytimg.com/vi/rh4IuW1fP6g/hqdefault.jpg)



